Are you looking for an answer to the topic “Does Lindlar’s catalyst reduce alkenes?“? We answer all your questions at the website Musicbykatie.com in category: Digital Marketing Blogs You Need To Bookmark. You will find the answer right below.
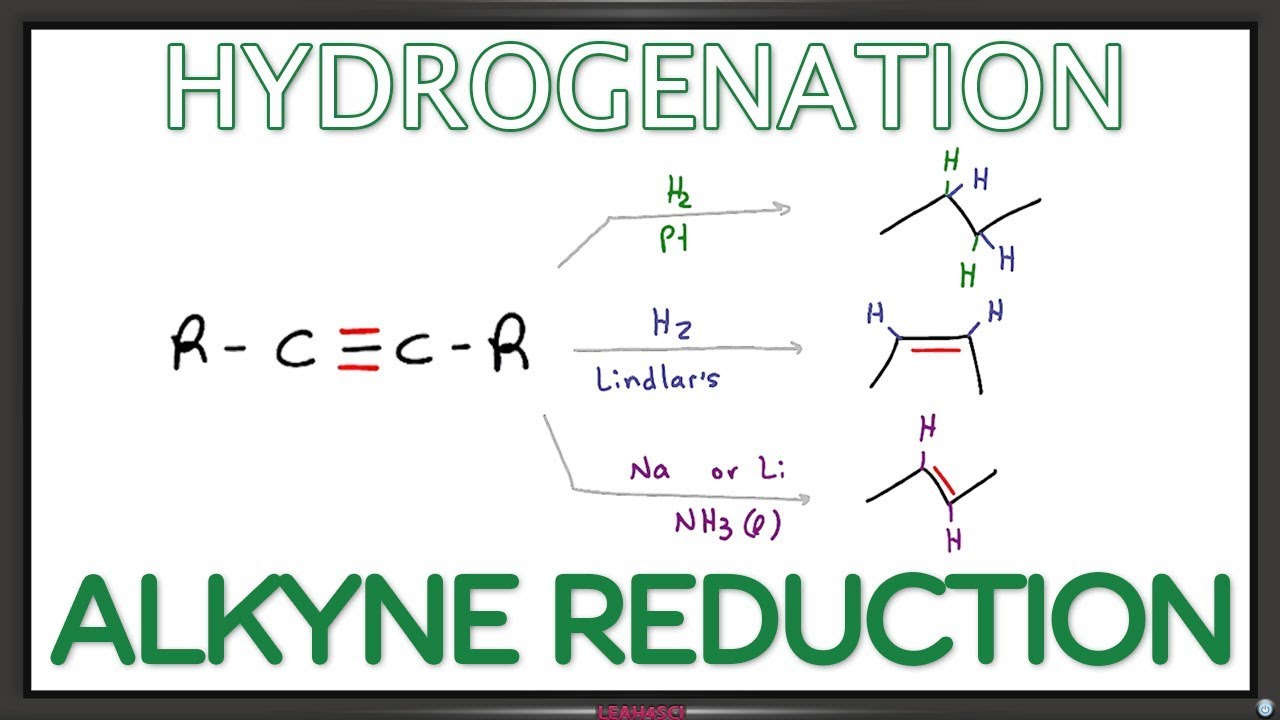
Lindlar’s catalyst is a palladium catalyst poisoned with traces of lead and quinoline, that reduce its activity such that it can only reduce alkynes, not alkenes.Lindlar’s Catalyst transforms an alkyne to a cis-alkene because the hydrogenation reaction is occurring on the surface of the metal. Both hydrogen atoms are added to the same side of the alkyne as shown in the syn-addition mechanism for hydrogenation of alkenes in the previous chapter.Alkenes undergo reduction by the addition of molecular hydrogen to give alkanes. Because the process generally occurs in the presence of a transition-metal catalyst, the reaction is called catalytic hydrogenation.

Table of Contents
What does Lindlar’s catalyst do to alkenes?
Lindlar’s Catalyst transforms an alkyne to a cis-alkene because the hydrogenation reaction is occurring on the surface of the metal. Both hydrogen atoms are added to the same side of the alkyne as shown in the syn-addition mechanism for hydrogenation of alkenes in the previous chapter.
Can alkenes be reduced?
Alkenes undergo reduction by the addition of molecular hydrogen to give alkanes. Because the process generally occurs in the presence of a transition-metal catalyst, the reaction is called catalytic hydrogenation.
Using Dissolving Metal Reduction or Lindlar’s Catalyst to turn alkynes to alkenes
Images related to the topicUsing Dissolving Metal Reduction or Lindlar’s Catalyst to turn alkynes to alkenes

Which reagent can reduce alkene?
An alkyne can be reduced to a Z (cis) alkene by “poisoning” the Pt catalyst so that the addition stops at the alkene; reagents that have been used are sulfur compounds such as barium sulfate, organic amines such as quinoline.
What is the function of Lindlar’s catalyst?
The catalyst is used for the hydrogenation of alkynes to alkenes (i.e. without further reduction into alkanes). The lead serves to deactivate the palladium sites, further deactivation of the catalyst with quinoline or 3,6-dithia-1,8-octanediol enhances its selectivity, preventing formation of alkanes.
Does Lindlar’s catalyst reduce terminal alkynes?
Lindlar’s catalyst is a palladium catalyst poisoned with traces of lead and quinoline, that reduce its activity such that it can only reduce alkynes, not alkenes.
How can we reduce alkynes to alkenes?
Alkynes can be reduced to trans-alkenes with the use of sodium dissolved in an ammonia solvent. An Na radical donates an electron to one of the P bonds in a carbon-carbon triple bond. This forms an anion, which can be protonated by a hydrogen in an ammonia solvent.
Which catalyst is suitable for preparing alkene?
Alkenes can be prepared from alkynes by carrying out hydrogenation in the presence of palletised charcoal. The charcoal which is used in this reaction has been moderately deactivated. Lindlar catalyst is palladium on calcium carbonate which has been deactivated by lead acetate to stop further hydrogenation.
See some more details on the topic Does Lindlar’s catalyst reduce alkenes? here:
Alkyne Reduction by Lindlar’s Catalyst or Na/NH3 – Chemistry …
In order to stop the reduction at alkenes, the reducing power of the H2 should be decreased and for this, a less active Pd catalyst is used in form of a Pd- …
Lindlar catalyst – Wikipedia
It is used for the hydrogenation of alkynes to alkenes (i.e. without further reduction into alkanes) and is named after its inventor Herbert Lindlar.
Reduction of Alkynes | MCC Organic Chemistry – Lumen …
Lindlar’s Catalyst: … Like alkenes, alkynes readily undergo catalytic hydrogenation, either to cis or trans alkenes, or to alkanes, depending on the reaction …
Alkyne Reactivity – MSU chemistry
Since alkynes adsorb more strongly to such catalytic surfaces than do alkenes, they preferentially occupy reactive sites on the catalyst. Subsequent transfer of …
Which catalyst is used for hydrogenation of alkenes?
Catalysts commonly used in alkene hydrogenation are: platinum, palladium, and nickel. The metal catalyst acts as a surface on which the reaction takes place.
Can LiAlH4 reduce alkenes?
Lithium aluminium hydride does not reduce simple alkenes or arenes. Alkynes are reduced only if an alcohol group is nearby. It was observed that the LiAlH4 reduces the double bond in the N-allylamides.
Which reagent can be used for reducing selectively alkynes to alkenes?
Selective reduction of alkenes and alkynes by the reagent lithium aluminum hydride-transition-metal halide.
Does NaBH4 reduce alkenes?
1 Answers. they cant reduce alkenes into alkanes because in a c=c there is no nucleophilic nature the H- ion needs a positive carbon to attack like in a functional group with oxygen the carbon has a slight possitive charge.
Alkyne Reduction – Hydrogenation, Lindar’s catalyst, Dissolving Metal Reduction – cis/trans alkenes
Images related to the topicAlkyne Reduction – Hydrogenation, Lindar’s catalyst, Dissolving Metal Reduction – cis/trans alkenes
Does LiAlH4 reduce double bond?
LiAlH4 reduces double bond in case of only when it is conjugation with phenyl ring but ”cinnamic acid” is an excepection because it has both carbonyl carbon as well as phenyl ring in its conjugation. LiAlH4 reduces double bond when it is in conjugation with phenyl but not in case of carbonyls.
What is Lindlar’s catalyst explain its role using but 2 YNE?
In hydrogenation using Lindlar’s catalyst, the hydrogen atoms are added on the same side of the molecule. This type of addition is known as syn addition. On treatment of 2-butyne with hydrogen gas and Lindlar’s catalyst, the major product obtained is cis-2-butene.
Why can a palladium Pd catalyst not be used to reduce an alkyne to an alkene?
Why can a palladium (Pd) catalyst not be used to reduce an alkyne to an alkene? Both π bonds will be reduced. The reduction of an alkyne will produce either a(n)_____ or a(n)______ as the product.
What is the product obtained when alkynes are subjected to hydrogenation in the presence of Lindlar’s catalyst?
Controlled hydrogenation of alkyne in presence of Lindlar’s catalyst yields cis product i.e., ‘cis’ alkene.
Can you reduce a terminal alkyne?
Alkynes can be reduced to trans-alkenes using sodium or lithium in liquid ammonia. The reaction, known as dissolving metal reduction, proceeds with an anti addition of hydrogen across the carbon–carbon triple bond to form the trans product.
Does Raney nickel reduce alkynes?
Applications. Raney nickel is used in a large number of industrial processes and in organic synthesis because of its stability and high catalytic activity at room temperature. It is typically used in the reduction of compounds that have multiple bonds, such as alkynes, alkenes, nitriles, dienes, aromatics and carbonyls …
Can you hydrogenate a terminal alkyne?
Hydrogenation of an Alkyne
Alkynes can be fully hydrogenated into alkanes with the help of a platinum catalyst. However, the use of two other catalysts can be used to hydrogenate alkynes to alkanes. These catalysts are: Palladium dispersed on carbon (Pd/C) and finely dispersed nickel (Raney-Ni).
What is Lindlar’s catalyst formula?
| PubChem CID | 131710810 |
|---|---|
| Molecular Formula | C4H6O4PbPd2 |
| Synonyms | Pd Lindlar catalyst |
| Molecular Weight | 538 |
| Component Compounds | CID 176 (Acetic acid) CID 23938 (Palladium) CID 5352425 (Lead) |
Which of these is also known as Lindlar’s catalyst?
Partially deactivated palladised charcoal (H2 /pd/CaCO3) is lindlar catalyst.
How do you turn an alkene into alkane?
To convert an alkene to an alkane, you must break the double bond by adding hydrogen to an alkene in the presence of a nickel catalyst, at a temperature of around 302 degrees Fahrenheit or 150 degrees Celsius, a process known as hydrogenation.
Catalytic Hydrogenation of Alkenes – Heterogeneous Catalysts
Images related to the topicCatalytic Hydrogenation of Alkenes – Heterogeneous Catalysts

How can we prepare alkene?
From alkyl halides: Alkenes are obtained by heating alkyl halides with alcoholic potash. Alcoholic potash is obtained by dissolving potassium hydroxide in alcohol. In this reaction, dehydrohalogenation takes place i.e. a single molecule of halogen acid is removed.
Can Methane be prepared by catalytic hydrogenation of alkene?
Methane cannot be prepared by catalytic hydrogenation of alkene.
Related searches to Does Lindlar’s catalyst reduce alkenes?
- does lindlar’s catalyst reduce alkenes
- what is catalytic hydrogenation of alkenes
- alkyne na nh3
- lindlars catalyst mechanism
- lindlar’s catalyst structure
- lindlar’s catalyst reaction
- what does lindlar’s catalyst do
- what catalyst is used in hydrogenation of alkenes
- what does lindlars catalyst do
- lindlar catalyst vs pdc
- which is lindlar’s catalyst
- lindlar catalyst vs pd/c
- can carboxylic acids be reduced by catalytic hydrogenation
- birch reduction of alkynes
- lindlar catalyst formula
- why lindlar’s catalyst is used in hydrogenation of alkynes
- reduction of alkynes
- alkyne to trans alkene
- lindlar’s catalyst mechanism
Information related to the topic Does Lindlar’s catalyst reduce alkenes?
Here are the search results of the thread Does Lindlar’s catalyst reduce alkenes? from Bing. You can read more if you want.
You have just come across an article on the topic Does Lindlar’s catalyst reduce alkenes?. If you found this article useful, please share it. Thank you very much.
