Are you looking for an answer to the topic “Does NaOH produce ions when dissolved in solution?“? We answer all your questions at the website Musicbykatie.com in category: Digital Marketing Blogs You Need To Bookmark. You will find the answer right below.
When sodium hydroxide (NaOH) dissolves in water, it separates into positively – charged sodium ions (cations) and negatively – charged hydroxide ions (anions).For example, when the base sodium hydroxide (NaOH) dissolves in water, it produces negative hydroxide ions and positive sodium ions (Na+). This can be represented by the chemical equation: NaOH H2O→ OH– + Na.In the context of the Brønsted Theory, a base can be thought of as an ionic compound that produces the hydroxide anion in solution. Thus, sodium hydroxide, NaOH, ionizes to form the sodium cation and the hydroxide anion (OH−).

Table of Contents
Does NaOH produce ions when dissolved in water?
For example, when the base sodium hydroxide (NaOH) dissolves in water, it produces negative hydroxide ions and positive sodium ions (Na+). This can be represented by the chemical equation: NaOH H2O→ OH– + Na.
Does NaOH form ions?
In the context of the Brønsted Theory, a base can be thought of as an ionic compound that produces the hydroxide anion in solution. Thus, sodium hydroxide, NaOH, ionizes to form the sodium cation and the hydroxide anion (OH−).
What Happens when Stuff Dissolves?
Images related to the topicWhat Happens when Stuff Dissolves?

What happens if NaOH reacts with water?
Sodium oxide reacts with water to produce sodium hydroxide.
What is formed when NaOH dissociates?
Hydroxide Ions
When NaOH dissolves into water, it dissociates into two ions: a positively charged sodium ion and a negatively charged hydroxide ion (OH-). The increased number of hydroxide ions in the solution increases the concentration of hydroxide ions in the water.
Does nacl dissociate in water?
When table salt, sodium chloride, dissolves in water, it dissociates into its respective cations and anions, Na+ and Cl–.
What ions do bases produce in water?
Acids produce hydronium ions in water, and bases produce hydroxide ions in water.
What ions does NaOH form in water?
When sodium hydroxide (NaOH) dissolves in water, it separates into positively – charged sodium ions (cations) and negatively – charged hydroxide ions (anions). These ions move around in the water, free and independent of each other, though cations tend to be surrounded more closely by anions and vice versa.
See some more details on the topic Does NaOH produce ions when dissolved in solution? here:
What Does Sodium Hydroxide React With? – Chemicals.ie
In water, sodium hydroxide is able to completely dissociate by breaking apart into sodium ions (Na+) and hydroxide ions (OH-).
The Effects of NaOH Concentration with H2O – Sciencing
When NaOH dissolves into water, it dissociates into two ions: a positively charged sodium ion and a negatively charged hydroxide ion (OH-). The …
Sodium hydroxide
It is very soluble in water with liberation of heat. It also dissolves in ethanol and methanol, though it exhibits lower solubility in these solvents than does …
What is Sodium Hydroxide? – Formula & Reactions – Study.com
Sodium hydroxide is also a strong base, which is a compound that splits apart in water to make many hydroxide (OH-) ions. When dissolved in …
How many ions are in NaOH?
Sodium Hydroxide is an ionic compound formed by two ions, Sodium Na+ and Hydroxide OH− . In order for these two polyatomic ions to bond the charges must be equal and opposite. Therefore, it take one +1 sodium ions to balance the one -1 hydroxide ion. This will make the formula for Sodium Hydroxide NaOH .
What ions exist in NaOH solution?
1 Answer. Sodium Hydroxide NaOH, is comprised of a positive sodium cation Na+ and a polyatomic hydroxide anion OH− .
Do bases dissociate in water?
Arrhenius argued that bases are neutral compounds that either dissociate or ionize in water to give OH– ions and a positive ion. NaOH is an Arrhenius base because it dissociates in water to give the hydroxide (OH–) and sodium (Na+) ions.
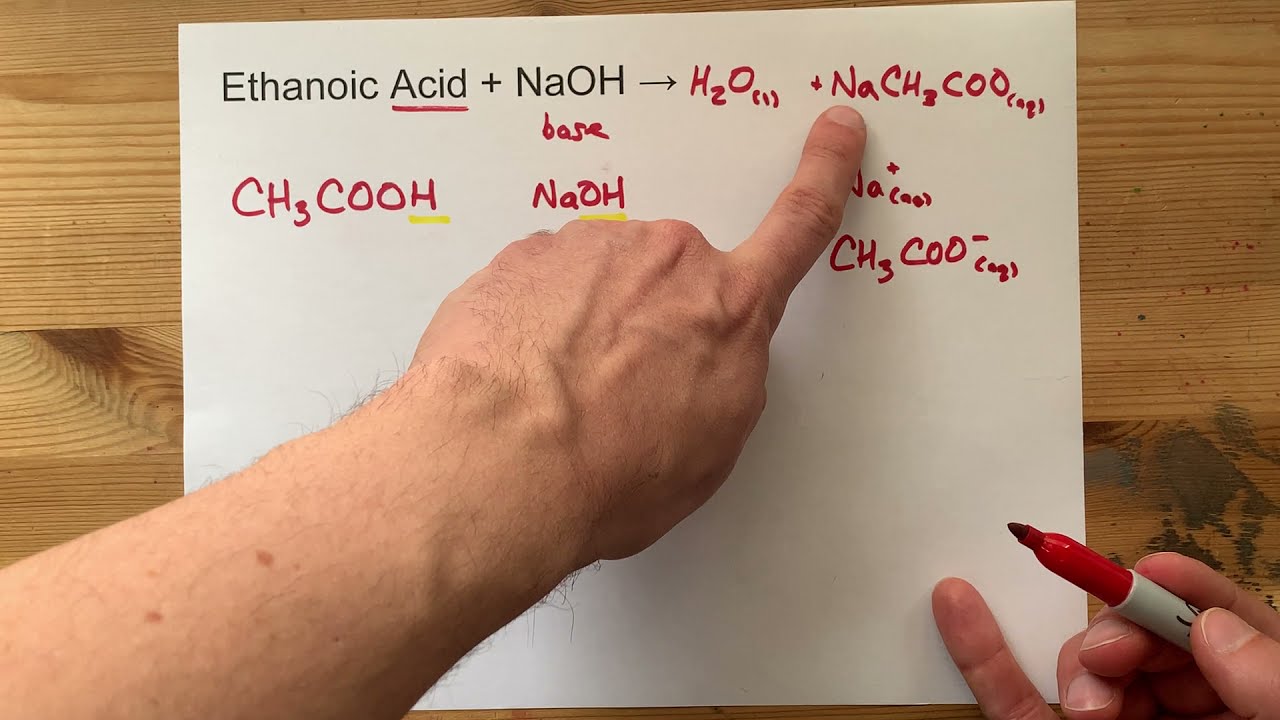
Ethanoic Acid + NaOH = ??
Images related to the topicEthanoic Acid + NaOH = ??
What is the product formed when sodium oxide reacts with water?
Reaction with water: Sodium oxide reacts exothermically with cold water to produce sodium hydroxide solution.
What happens when Na2O is dissolved in water?
Na2O reacts violently when exposed to water, forming sodium hydroxide in the process.
What ions are formed when sodium hydroxide dissolves in water quizlet?
When sodium hydroxide dissolves in water, it gives off hydroxide ions. This increases the concentration of hydroxide ions found in the aqueous solution.
When water dissociates what ions are released?
Water is a neutral liquid. When it dissociates it splits into its ions which are hydronium ion (H30) and hydroxide ion (OH).
Is Naoh soluble in water?
What happens when NaCl dissolves in solution?
Water molecules pull the sodium and chloride ions apart, breaking the ionic bond that held them together. After the salt compounds are pulled apart, the sodium and chloride atoms are surrounded by water molecules, as this diagram shows. Once this happens, the salt is dissolved, resulting in a homogeneous solution.
When NaCl is dissolved in water the sodium ions become?
Hence, we can conclude that when NaCl is dissolved in water, the sodium ion becomes hydrated. Hence, the correct option for this question is D that is hydrated.
What ions do bases produce in solution?
In chemistry, acids and bases have been defined differently by three sets of theories. One is the Arrhenius definition, which revolves around the idea that acids are substances that ionize (break off) in an aqueous solution to produce hydrogen (H+) ions while bases produce hydroxide (OH–) ions in solution.
Is NaOH an acid or base?
Sodium hydroxide (Na OH), also known as lye or caustic soda, is a caustic metallic base. An alkali, caustic soda is widely used in many industries, mostly as a strong chemical base in the manufacture of pulp and paper, textiles, drinking water, and detergents.
Calculating Ion Concentrations in Solution
Images related to the topicCalculating Ion Concentrations in Solution

What kind of ions are released in a basic solution?
A base is a substance that releases hydroxyl ions (OH–) when in solution.
What is the product of NaOH?
Sodium hydroxide is also known as lye or soda , or caustic soda.
Related searches to Does NaOH produce ions when dissolved in solution?
- what does naoh do in a reaction
- what is a solution called that has more oh ions h ions
- substances that release hydroxide ions when dissolved in water are called
- what does naoh react with
- what ions do bases produce in water
- what is a solution called that has more oh-ions h+ ions
- does naoh produce ions when dissolved in solution is
- does naoh produce ions when dissolved in solution is called
- naoh reagent organic chemistry
- sodium hydroxide and water exothermic or endothermic
- does naoh produce ions when dissolved in solution water
- what ions are formed when sodium hydroxide dissolves in water quizlet
Information related to the topic Does NaOH produce ions when dissolved in solution?
Here are the search results of the thread Does NaOH produce ions when dissolved in solution? from Bing. You can read more if you want.
You have just come across an article on the topic Does NaOH produce ions when dissolved in solution?. If you found this article useful, please share it. Thank you very much.
